 What are the existing levels of uranium in the environment? The table below shows the average energies per transformation emitted by U-238, U-235 and U-234.Īverage Energy Emitted Per Transformation (MeV Bq -1)Ģ. The emission of beta particles and gamma radiations are low. Isotopes of natural uranium decay by emitting mainly alpha particles. the activity of each of the radioactive progeny is equal to the activity of the uranium parent isotope) with their radioactive decay products. In nature, uranium isotopes are typically found in radioactive equilibrium (i.e. The activity concentration arising solely from the decay of the uranium isotopes (U-234, U-235 and U-238) found in natural uranium is 25.4 Bq per mg. An activity of one becquerel (Bq) means that on average one disintegration takes place every second. It is expressed in the table in becquerels (Bq) per milligram (1 milligram, mg, = 0.001 grams). The specific activity is the activity per unit mass of a particular radionuclide and is used as a measure of how radioactive a radionuclide is. The half life of a radioactive isotope is the time taken for it to decay to half of its original amount of radioactivity. 
The table below shows the fraction by weight of the three isotopes in any quantity of natural uranium, their half lives, and specific activity. Other isotopes that cannot be found in natural uranium are U-232, U-233, U-236 and U-237. In its natural state, it consists of three isotopes (U-234, U-235 and U-238). The International Atomic Energy Agency (IAEA) defines uranium as a Low Specific Activity material. It is so dense a small 10-centimetre cube would weigh 20 kilograms. Like tungsten it is very dense, about 19 grams per cubic centimetre, 70% more dense than lead. In its pure form it is a silver-coloured heavy metal, similar to lead, cadmium and tungsten. Compared to the γ energy spectrum method, the relative error of each measurement result was between 1.11% and 1.66%.Uranium (chemical symbol U) is a naturally occurring radioactive element. To verify the precision and accuracy of this method, 5 tests using each working curve were conducted for simulated samples with a 235U abundance of 72.2%, and the relative standard deviation (RSD) of all tests was less than 2%. 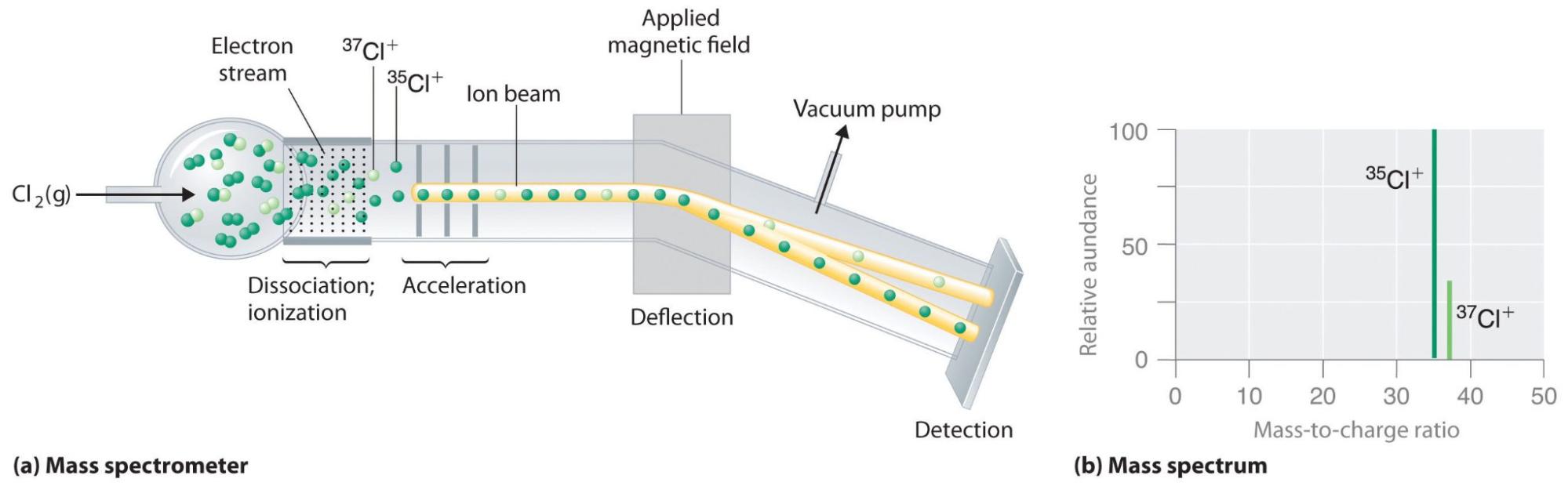
Taking the fission-yield ratios of specific fission products, such as Y¯88Rb/Y¯107Rh, Y¯88Rb/Y¯135I, Y¯88Rb/Y¯104Tc and Y¯92Sr/Y¯104Tc as the subjects of research, the relation curves between the average yield ratios and the 235U isotopic abundance as well as the expressions (Y¯i/Y¯j=f(H0)) for the average yield ratio (Y¯i/Y¯j) as a function of the 235U isotopic abundance (H0) were obtained. The exponential relation between different abundances of 235U and the average fission-yield ratios of specific fission products was employed in this method. 
Based on neutron activation analysis, a new method was presented for the determination of uranium isotopic abundance, in which fast neutrons generated by T (d, n) 4He were used to induce fission in uranium samples. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
